Cytology is the most common and immediately informative diagnostic tool in the practice of dermatology to achieve a rapid, convenient and inexpensive indication of whether a patient has a skin infection, what type of infections (yeast vs. bacteria) and what type of inflammatory cells are present. To perform ear cytology there is no need for specialist equipment apart from cotton buds, slides, staining and a good microscope.
This short article will focus on sample collection techniques and the identification of infectious organisms, inflammatory cells, changes in keratinocyte morphology and artifacts that may be encountered. It does not include sampling technique from nodules, or cutaneous neoplasms, as their interpretation requires additional expertise and specialized training, and is beyond the
scope of this article. To perform skin cytology there is no need for specialist equipment apart from adhesive tape, slides, staining fluid and a good microscope.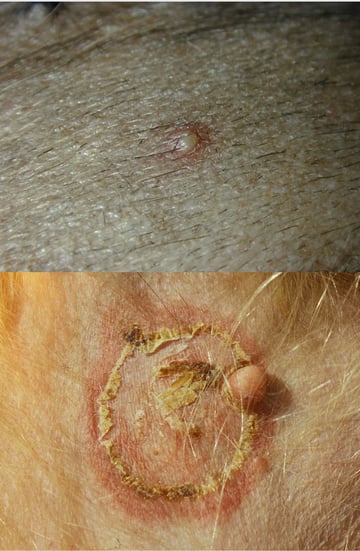 Cytology is normally done from skin lesions like pustules (fig 1), edges of collarettes (fig 2), plaques, crusts, ulcers, non-healing wounds or fistulae, as well as skin folds and interdigital spaces.
Cytology is normally done from skin lesions like pustules (fig 1), edges of collarettes (fig 2), plaques, crusts, ulcers, non-healing wounds or fistulae, as well as skin folds and interdigital spaces.
It may help to confirm the presence and evaluate the type of an infection/ inflammatory response and acantholysis. It eventually helps with deciding if a swab for culture and sensitivity, or further diagnostics tests are needed.
The cytological findings are also invaluable in determining the treatment to use and monitoring the response to the therapy.
The technique of choice depends from the site to sample:
Glass Slide Impressions
Impressions may be taken from any exudative lesion. A pustule is sampled by opening its top with a fine needle before a few impressions are taken directly onto a glass slide. Moving the slide slightly before each impression will avoid too thick a build-up of material. Sampling the edge of a collarette may yield more microorganisms than sampling the centre of the lesion. Where you
have a crust, it is better to sample the skin surface or the area beneath the crust. Impressions of the underside of a crust can be diagnostically valuable, particularly in cases of suspected pemphigus foliaceus. The material discharging from a fistula should be collected by pressing the skin around the fistula or inserting a cotton swab inside the fistula.
Care should be taken when obtaining direct impressions to prevent cellular damage, which will render the cytology un- interpretable. Where a direct impression is not possible, material may be transferred from a lesion to the slide using a cotton bud.
Acetate Strips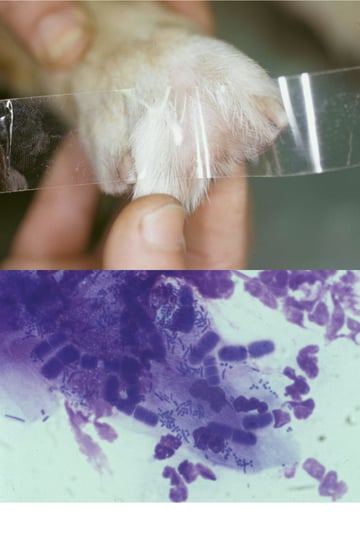
Transparent acetate tape is used to obtain a sample from dry lesions that will not stick to a glass slide easily, or to obtain samples from areas (skin folds and interdigital spaces, fig 3) that are difficult to get to with a glass slide.
A strip of tape about 50 per cent longer than a microscope slide is used and the middle of the sticky side of the tape is pressed firmly against the area several times to pick up epidermal debris and obtain samples. Each end of the tape is then attached to one end of a microscope slide, forming a loop, and stained. Then wrapping the tape around both ends of the slide holds it firmly in position and facilitates microscopic examination under oil immersion.
With samples such as pus, the slide is first air-dried and then stained. The easiest stain to use in-house is a commercial modified Wright’s stain like Diff Quik. Note that bacteria and yeasts always stain blue if using the Diff-Quik staining. Similarly, acetate tape strip samples are stained with Diff Quik but just using the red and blue dyes. Use of the alcohol fixative would dissolve the glue of the tape.
During the microscopical examination it is important to keep the light condenser up when examining a cytology sample. Cytological preparations should first be scanned under low power for an area of evenly distributed cells, one layer thick.
The use of a x100 oil immersion objective is then recommended for detailed examination of cellular morphology.
The most common types of organisms encountered in cutaneous cytology are yeast and bacteria. The most common yeast organism found on samples from dogs and cats is Malassezia pachydermatis, although Candida spp can also be found on the skin. The variety of bacteria that can be found on cutaneous cytology is vast. However, Staphylococcus spp. is the most common bacteria isolated from infections of skin in dogs and cats. Another bacterium occasionally encountered is Simonsiella spp (fig 4).
These are harmless, very large filamentous Gram-negative saprophytes that inhabit the oral cavity of a variety of warm blooded vertebrates. They are usually found in small numbers and are not intracellular (although they can be adherent to keratinocytes).
These organisms are not known to be pathogenic; however, when found on areas not associated with the oral cavity, such as the interdigital spaces, their presence is likely associated with pruritus.
Dermatophyte and other fungal organisms can also be found on cytology. Dermatophyte spores often appear as round spheres about twice the size of coccoid bacteria.
Identifying inflammatory cells on cytology can give a clue as to the pathogenesis of the lesion. Presence of degenerate neutrophils with intracellular bacteria is most often indicative of a pyoderma. If well-preserved neutrophils are seen then conditions such as pemphigus foliaceus may be considered. At times, the force that is required to obtain an exfoliative sample
can rupture any present inflammatory cells, especially neutrophils. When this occurs, the nuclear material from these cells often streaks the slide and can be identified on low power. The presence of eosinophils indicates inflammation secondary to parasites,
hypersensitivity reactions or eosinophilic granuloma in cats.
Other common inflammatory cells recognized are macrophages and lymphocytes. These are usually seen from more chronic or deeper lesions. Macrophages are efficient phagocytic cells and often contain effete neutrophils, red blood cells, bacteria, fungal elements and even intracellular parasites such as Leishmania spp. organisms and others.
Epithelial cells encountered normally on exfoliative cytology are corneocytes (non-nucleated keratinocytes). Keratinocytes are a normal finding; however, in diseased skin, keratinocyte morphology often has diagnostic value. Nucleated keratinocytes are normally found in samples from mucous membranes or when the surface of the skin was scraped before cytology. When found from surface skin samples it is suggestive of abnormal turnover of the epithelial layers or erosion of the cornified layers. These keratinocytes often contain keratohyaline granules and should not be confused with bacteria. Instead of appearing as flat sharply angulated large cells, some keratinocytes or corneocytes can appear rolled or more spindle shaped on routine cytology because of folding and rolling during the sampling and fixation process. Acantholytic cells are lower layer keratinocytes that have lost their adhesion to neighbouring keratinocytes. They are most commonly found in pemphigus foliaceus lesions, but can also be seen with deep furunculosis secondary to conditions such as demodicosis and dermatophytosis. Acantholytic cells are large, round cells with a central nucleus. They often stain with a dark blue periphery that may indicate antibody coating.
In cases of pemphigus, they are often found in rafts and among well preserved neutrophils and/or eosinophils. In cases of demodicosis or dermatophytosis, acantholytic cells are usually sparse and found among degenerate inflammatory cells. Artifacts are often encountered on cytology slides. They can often be confused for significant findings. Many exudative samples
have a markedly proteinaceous background, which often looks light purple or pink with fine darker staining granules that can be mistaken for small bacteria. Melanin granules can also be mistaken for rod-shaped bacteria such as Pseudomonas spp.
However, melanin granules are most often found in and around keratinocytes, stain a refractory brown to black colour and are very even in size.
It is not uncommon during the Spring/Summer months to find, especially from samples taken from the interdigital spaces, vegetable pollens. They come in multiple shapes and sizes and are often confused for fungal elements.
Conclusions
In a given month at a dermatology referral practice, cytological samples are taken from approximately 80% of patients presented, comprising about 10 to 20% of the clinic’s total income!
Cutaneous cytology is inexpensive, highly informative and easily self- taught. By making cytology part of everyday practice, diagnostics and treatments become more proficient and case management is immediately more rewarding. The ultimate result is significantly happier patients and clients.
 Global English
Global English

 Deutschland
Deutschland
 UK
UK